
Chemistry, 13.12.2020 04:40 jadeochoa4466
For the following reaction, calculate how many moles of each product are formed when 4.05 g of water is used. 2H20 →2H2 + O2

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1


Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1
You know the right answer?
For the following reaction, calculate how many moles of each product are formed when 4.05 g of water...
Questions in other subjects:


Physics, 11.07.2019 19:30

Mathematics, 11.07.2019 19:30




Mathematics, 11.07.2019 19:30


Mathematics, 11.07.2019 19:30

Mathematics, 11.07.2019 19:30

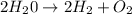
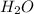 is used to form 2 moles of
is used to form 2 moles of 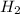 and 1 mole of
and 1 mole of 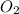
 , 1 mole of
, 1 mole of  , and 1 mole of
, and 1 mole of 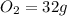 .
.

