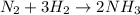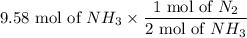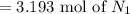Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, ElizabethF
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 23.06.2019 01:30, Sonicawesomeness
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2

Chemistry, 23.06.2019 06:00, girlwonder326
+= + + balance the equation on coefficients, ex. 1,1,1
Answers: 2

Chemistry, 23.06.2019 06:00, mirzakasumovic8926
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2
You know the right answer?
For the reaction N2 + 3H2 - 2NH3, how many moles of nitrogen are required to produce 9.58 mol of amm...
Questions in other subjects:





Mathematics, 18.02.2022 20:20

English, 18.02.2022 20:20


English, 18.02.2022 20:20

Mathematics, 18.02.2022 20:20

English, 18.02.2022 20:20