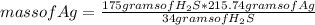Chemistry, 12.12.2020 17:10 haleyzoey7
My grandmother left me some silver years ago. I opened the box containing the silver and it had tarnished. Using the equation below, if I had 300 g of silver that had tarnished and 175g H2S, which would be the limiting reactant?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, zitterkoph
Which of the following mining methods disrupts the sea floor?
Answers: 1

Chemistry, 21.06.2019 22:30, 20heldmadison
Hot air balloons float in the air because of the difference in density between cold and hot air. in this problem, you will estimate the minimum temperature the gas inside the balloon needs to be, for it to take off. to do this, use the following variables and make these assumptions: the combined weight of the pilot basket together with that of the balloon fabric and other equipment is w. the volume of the hot air inside the balloon when it is inflated is v. the absolute temperature of the hot air at the bottom of the balloon is th (where th> tc). the absolute temperature of the cold air outside the balloon is tc and its density is ďc. the balloon is open at the bottom, so that the pressure inside and outside the balloon is the same. as always, treat air as an ideal gas. use g for the magnitude of the acceleration due to gravity.
Answers: 1


Chemistry, 22.06.2019 11:00, daniel1480
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced. be sure your answer has the correct number of significant digits.
Answers: 2
You know the right answer?
My grandmother left me some silver years ago. I opened the box containing the silver and it had tarn...
Questions in other subjects:

Mathematics, 12.02.2021 05:20


Mathematics, 12.02.2021 05:20

Mathematics, 12.02.2021 05:20

History, 12.02.2021 05:20




English, 12.02.2021 05:20