
Chemistry, 12.12.2020 16:50 yarbor800592
Hydrazine reacts with oxygen according to the following equation: N2H4(g) +O2(g) → N2(g) + 2 H2O(l) How many L of N2, measured at 34.9 °C and 755.08 torr, will be produced at the same time that 914.894 g of H2O is produced?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, dyanaycooper13
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1


Chemistry, 22.06.2019 16:30, Kathryn014
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

You know the right answer?
Hydrazine reacts with oxygen according to the following equation: N2H4(g) +O2(g) → N2(g) + 2 H2O(l)...
Questions in other subjects:



Mathematics, 20.09.2020 16:01




Mathematics, 20.09.2020 16:01


History, 20.09.2020 16:01

Health, 20.09.2020 16:01

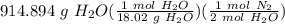 = 25.3955 mol N₂
= 25.3955 mol N₂

