
A concept map for four types of intermolecular forces and a certain type of bond is shown.
Which of the following correctly identifies the intermolecular force represented by A and compares its strength relative to the intermolecular force represented by b.
1. A represents London dispersion forces, which are weaker than the force represented by b
2. A represents hydrogen bonding, which is weaker than the force represented by b
3. A represents London dispersion forces, which are stronger than the force represented by b
4. A represents hydrogen bonding, which is stronger than the force represented by b
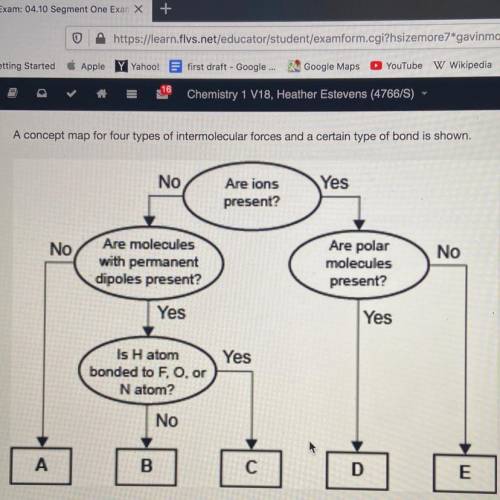

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, NREYESLDS2806
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1


Chemistry, 22.06.2019 23:30, znewkirk4741
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3
You know the right answer?
A concept map for four types of intermolecular forces and a certain type of bond is shown.
Which of...
Questions in other subjects:


History, 15.12.2019 00:31


Biology, 15.12.2019 00:31

Chemistry, 15.12.2019 00:31




English, 15.12.2019 00:31



