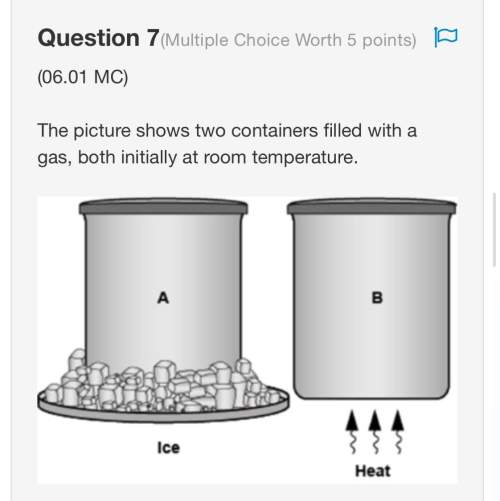
Chemistry, 10.12.2020 16:10 mathlady12
A flask of fixed volume contains 1.0 mole of gaseous carbon dioxide and 88 g of solid carbon dioxide. The original pressure and temperature in the flask is 1.0 atm and 300. K. All of the solid carbon dioxide sublimes. The final pressure in the flask is 2.5 atm. What is the final temperature?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:30, tiffanyhmptn
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 23.06.2019 00:00, ahmedeldyame
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1


Chemistry, 23.06.2019 11:40, brendonvernon8
Which of the following would have the lowest average kinetic energy
Answers: 1
You know the right answer?
A flask of fixed volume contains 1.0 mole of gaseous carbon dioxide and 88 g of solid carbon dioxide...
Questions in other subjects:







Mathematics, 07.03.2020 03:57