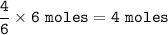Consider the following balanced equation:
P4(s) + 6F2(e)
4PF3(g)
If 1.25 moles of P4(s)...

Chemistry, 09.12.2020 17:00 tonydeanfbg8706
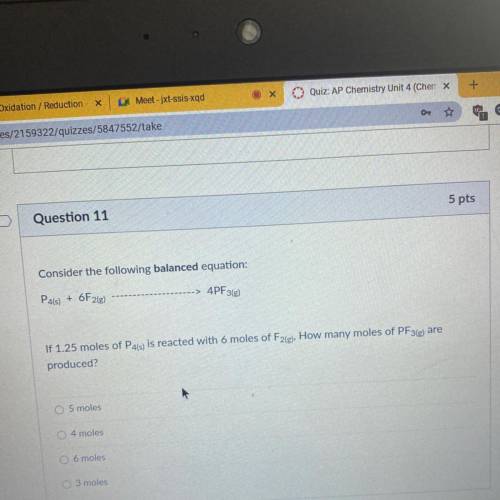
Consider the following balanced equation:
P4(s) + 6F2(e)
4PF3(g)
If 1.25 moles of P4(s) is reacted with 6 moles of F2(g), How many moles of PF3(e) are
produced?
O 5 moles
4 moles
O 6 moles
O 3 moles

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:10, glitterpanda2468
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2


Chemistry, 23.06.2019 01:00, ZaNiyahlove4711
Which of the following is the molecular formula for a simple sugar? a. cooh b. h2o c. oh d. c6h12o6
Answers: 1

Chemistry, 23.06.2019 04:00, anonymous1813
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2
You know the right answer?
Questions in other subjects:

Chemistry, 30.06.2019 03:30



Mathematics, 30.06.2019 03:30

Mathematics, 30.06.2019 03:30

English, 30.06.2019 03:30

Mathematics, 30.06.2019 03:30

Mathematics, 30.06.2019 03:30

History, 30.06.2019 03:30

History, 30.06.2019 03:30