
Chemistry, 05.12.2020 01:00 maria241432
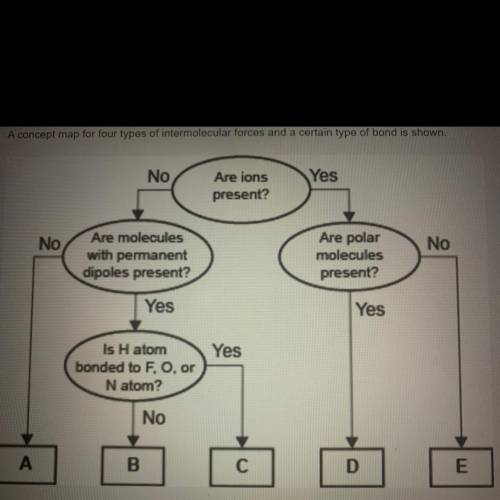
Pls help me, I’m not too sure! A concept map for four types of intermolecular forces and a certain type of bond is shown.
Compare the relative strength of the two forces B and C. Explain how you determined this comparison by identifying the forces.
(This is a free response question, not a multiple choice.)

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:30, tbeck225
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1


Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
You know the right answer?
Pls help me, I’m not too sure! A concept map for four types of intermolecular forces and a certain t...
Questions in other subjects:



Mathematics, 28.02.2021 20:50

Biology, 28.02.2021 20:50


Mathematics, 28.02.2021 20:50


Mathematics, 28.02.2021 20:50


Business, 28.02.2021 20:50



