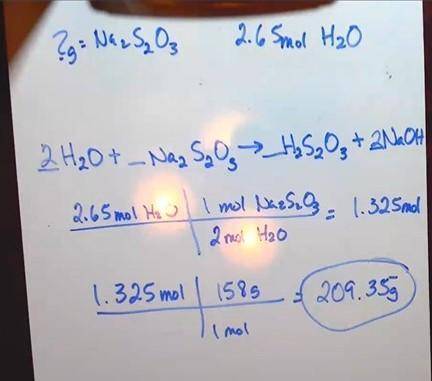Chemistry, 04.12.2020 22:10 aljalloh94
PLS HELP! I CAN"T DO THIS! I AM ABOUT TO CRY COZ NO ONE WILL ANSWER TnT
Chlorine is used by textile manufacturers to bleach cloth. Excess chlorine is destroyed by its reaction with sodium thiosulfate, Na 2 S 2 O 3 :
Na 2 S 2 O 3(aq) + 4Cl 2(g) + 5H 2 O (aq) 2NaHSO 4(aq) + 8HCl (aq)
1. How many grams of Na 2 S 2 O 3 are needed to react with 2.65 mol of H 2 O? (3
marks)
2. How many mol of HCl can form from 25.2 mol of Na 2 S 2 O 3 ? (2 marks)
3. How many Liters of Cl 2 are required to produce 15.7 moles of NaHSO 4 ? (2
marks)
4. How many molecules of HCl can form from 4.92 grams of H 2 O? (3 marks)
I also need the steps. Please and thank you!

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, pressure772
Which is a character of nuclear fusion but not nuclear fission
Answers: 3


Chemistry, 22.06.2019 15:30, alaf05160
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks. energy was destroyed inside the blocks. energy was absorbed into the blocks from outside the system. energy was transferred from the warmer block to the cooler block.
Answers: 2
You know the right answer?
PLS HELP! I CAN"T DO THIS! I AM ABOUT TO CRY COZ NO ONE WILL ANSWER TnT
Chlorine is used by textile...
Questions in other subjects:



Mathematics, 26.09.2019 13:50

Social Studies, 26.09.2019 13:50

Mathematics, 26.09.2019 13:50

Biology, 26.09.2019 13:50

Mathematics, 26.09.2019 13:50


English, 26.09.2019 13:50