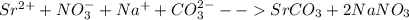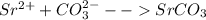Strontium compounds are often used in flares because their flame color is bright red. One industrial process to produce low-solubility strontiun) compounds (that are less affected by getting wet) involves the reaction of aqueous solutions of strontium nitrate and sodium carbonate. Write the balanced molecular equation, the total ionic equation and the net ionic equation for this reaction.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, alexandroperez13
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 21.06.2019 23:30, huangjianhe135
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 06:00, tddreviews
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1
You know the right answer?
Strontium compounds are often used in flares because their flame color is bright red. One industrial...
Questions in other subjects:




Computers and Technology, 31.08.2019 04:00

History, 31.08.2019 04:00

History, 31.08.2019 04:00

History, 31.08.2019 04:00

Social Studies, 31.08.2019 04:00

History, 31.08.2019 04:00