
Chemistry, 03.12.2020 14:00 Lakenwilliams1250
If in the reaction below 32 grams of C2H6 produces 44 grams of CO2, what is the % yield? 2C2H6 + 7O2 = 4CO2 + 6H2O imagine the = is an arrow

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, speris1443
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 14:00, claudia122752
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 18:00, liddopiink1
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3
You know the right answer?
If in the reaction below 32 grams of C2H6 produces 44 grams of CO2, what is the % yield? 2C2H6 + 7O2...
Questions in other subjects:






Mathematics, 24.02.2021 01:30


Mathematics, 24.02.2021 01:30


Mathematics, 24.02.2021 01:30

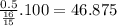 %
%

