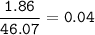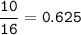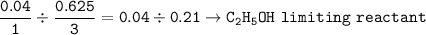Consider the following unbalanced equation:
C2H5OH(g) + O2(g) → CO2(g) + H2O(l)
1.86 g of et...

Chemistry, 03.12.2020 14:00 Seumas9307
Consider the following unbalanced equation:
C2H5OH(g) + O2(g) → CO2(g) + H2O(l)
1.86 g of ethanol reacts with 10.0 g of oxygen. What is the total volume of gas present (in L) after the reaction is complete, assuming the reaction takes place at 1.00 atm and 25°C?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, elizabethprasad2
How many grams of n2h4 will be consumed by 23 g of n2o4
Answers: 1

Chemistry, 22.06.2019 20:30, camerondillonn
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1

Chemistry, 22.06.2019 21:50, BookandScienceNerd
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
Questions in other subjects:

Mathematics, 28.10.2019 18:31



English, 28.10.2019 18:31





Mathematics, 28.10.2019 18:31