Chemistry, 02.12.2020 17:10 BrianKeokot7700
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liquid water at 298 K. b) Calculate the standard reaction entropy for the hydrolysis of liquid water to form oxygen and hydrogen gas. c) Do your results in (a) and (b) agree with the general rule that reaction entropies are positive if there is a net formation of gas in a reaction, and negative if the is a net reduction of gas?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:40, loveoneonly9153
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1


Chemistry, 22.06.2019 11:00, Usman458
The twister and runaway train are two coasters at the same amusement park. both coasters start at the same height. the coaster for the twister is twice the mass of the coaster for the runaway train. which roller coaster has greater gravitational potential energy at the start of the ride?
Answers: 1
You know the right answer?
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liqui...
Questions in other subjects:

Mathematics, 22.05.2020 15:57



English, 22.05.2020 15:57

Mathematics, 22.05.2020 15:57


Mathematics, 22.05.2020 15:57

Mathematics, 22.05.2020 15:57


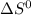 = - 242.2J/K.mol
= - 242.2J/K.mol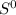 .
. 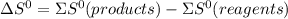
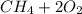 ⇒
⇒ 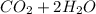
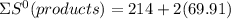 = 353.8
= 353.8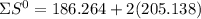 = 596.54
= 596.54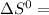 353.8 - 596.54
353.8 - 596.54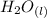 ⇒
⇒ 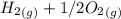
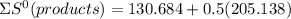 = 233.253
= 233.253