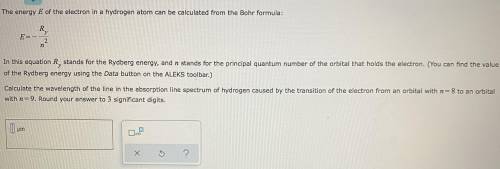
Chemistry, 02.12.2020 17:00 AreYouMyNewDad
g In this equation stands for the Rydberg energy, and stands for the principal quantum number of the orbital that holds the electron. (You can find the value of the Rydberg energy using the Data button on the ALEKS toolbar.) Calculate the wavelength of the line in the emission line spectrum of hydrogen caused by the transition of the electron from an orbital with to an orbital with . Round your answer to significant digits.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, tbiles99
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1



Chemistry, 23.06.2019 03:30, tamariarodrigiez
Which of the following describes the entropy change as a solution is made from a liquid and solid
Answers: 1
You know the right answer?
g In this equation stands for the Rydberg energy, and stands for the principal quantum number of the...
Questions in other subjects:

History, 23.12.2020 18:10


Mathematics, 23.12.2020 18:10

Mathematics, 23.12.2020 18:10

Mathematics, 23.12.2020 18:10

Mathematics, 23.12.2020 18:10


Biology, 23.12.2020 18:10

Chemistry, 23.12.2020 18:10