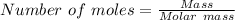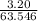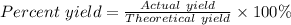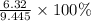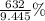Chemistry, 02.12.2020 01:00 angelica7773
Suppose 3.20 g of copper are reacted with excess nitric acid according to the given equation, and 6.32 g Cu(NO3)2 product are obtained.
Cu(s) + 4 HNO3 (aq) --> Cu(NO3)2 (aq) + 2 NO2 (g) + 2 H2O(l)
What is the theoretical yield of Cu(NO3)2? In g
What is the percent yield of Cu(NO3)2? In %

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, chloe081
We just started a new lesson in chemistry and everyone hates it and i dont get it one bit. i hate school. h el p. balanced equationc3h8+5o2-> 3co2+4h2o1.) if you start with 14.8g of propane(c3h8) and 3.44g of oxygen, which is the limiting reactant -check my answer 2.)what mass of excess reagent is left over? 3.)what mass of carbon dioxide can be made? 4.)what mass of water is produced?
Answers: 2

Chemistry, 23.06.2019 02:30, elyzarobertson
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
You know the right answer?
Suppose 3.20 g of copper are reacted with excess nitric acid according to the given equation, and 6....
Questions in other subjects:


Mathematics, 17.04.2020 00:04

Mathematics, 17.04.2020 00:04

Mathematics, 17.04.2020 00:04



Chemistry, 17.04.2020 00:04


Arts, 17.04.2020 00:04