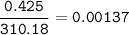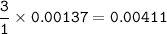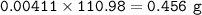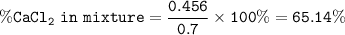Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, tiwaribianca475
What volume of a 2.00 m stock solution of naoh is needed to prepare 150. ml of 0.40 m solution?
Answers: 2


Chemistry, 22.06.2019 05:30, nuclearfire278
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease. correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1
You know the right answer?
A 0.700 g unknown mixture with CaCl2 reacts with aqueous sodium phosphate to give 0.425 g Ca3(PO4)2....
Questions in other subjects:




Mathematics, 10.12.2020 02:00





English, 10.12.2020 02:00