
Chemistry, 30.11.2020 01:10 Robinlynn228
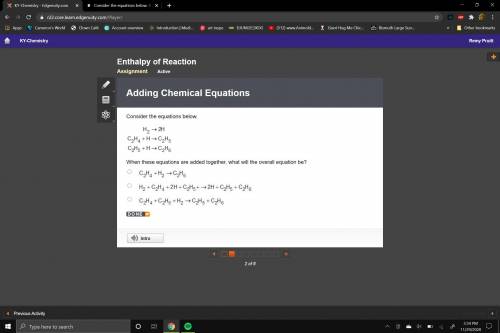
Consider the equations below.
3 equations. 1: upper H subscript 2 right arrow 2 upper H.
2: upper C subscript 2 upper H subscript 4 plus upper H right arrow upper C subscript 2 upper H subscript 5.
3: upper C subscript 2 upper H subscript 5 plus upper H right arrow upper C subscript 2 upper H subscript 6.
When these equations are added together, what will the overall equation be?
Upper C subscript 2 upper H subscript 4 plus upper H subscript 2 right arrow upper C subscript 2 upper H subscript 6.
Upper H subscript 2 plus upper C subscript 2 upper H subscript 4 plus 2 upper H right arrow 2 upper H plus upper C subscript 2 upper H subscript 5 plus upper C subscript 2 upper H subscript 6.
Upper C subscript 2 upper H subscript 4 plus upper C subscript 2 upper H subscript 5 plus upper H subscript 2 right arrow upper C subscript 2 upper H subscript 5 plus upper C subscript 2 upper H subscript 6.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, Blaise2653
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1


Chemistry, 22.06.2019 16:00, winnie45
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2
You know the right answer?
Consider the equations below.
3 equations. 1: upper H subscript 2 right arrow 2 upper H.
Questions in other subjects:

Mathematics, 24.05.2021 08:30


English, 24.05.2021 08:30

Mathematics, 24.05.2021 08:30

Mathematics, 24.05.2021 08:30




Mathematics, 24.05.2021 08:30



