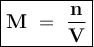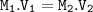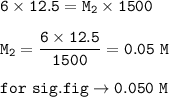Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:30, terrancebest
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2


Chemistry, 22.06.2019 09:00, wkalpakchi
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1
You know the right answer?
12.5 mL of 6.0 M KCl is diluted to make a 1.5 L solution. The molarity of the dilution solution is _...
Questions in other subjects:








Mathematics, 26.10.2020 16:40

Social Studies, 26.10.2020 16:40

Chemistry, 26.10.2020 16:40