
Chemistry, 28.11.2020 07:30 xmanavongrove55
25 points , hi! please look at the attachment for the question, I'm having a hard time because all I have to do is solve through those 2 problems but I'm not sure if I solve the fractions first then multiply that result by 100 or what. I asked my teacher which she helped a little but I don't think she understood where I was lost. If you can help I would really appreciate it. Thank you.
This was the set up question:
4. If 100.0g of nitrogen gas (N2) is reacted with 100.0g of hydrogen gas (H2) to form NH3. What is the limiting and excess reactants?
Hint: Convert grams to moles for each reactant and then convert to moles of NH3. You need your balanced equation from answer 1 to determine the mole relationship between each reactant and the product NH3. Use the periodic table to determine the molar mass of all chemical formulas. Fill in the “?” blanks below to show your work.
and in the screenshot it has everything I'm working with and the conclusions I need to draw from it, I can draw the conclusions just fine on my own but I need help solving.


Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 06:00, Sarahinator04
0.09 moles of sodium sulfate in 12 ml of solution
Answers: 3

Chemistry, 22.06.2019 12:30, poopybutt541
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 19:00, andrecoral105
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1
You know the right answer?
25 points , hi! please look at the attachment for the question, I'm having a hard time because all I...
Questions in other subjects:

Mathematics, 14.10.2020 18:01




Social Studies, 14.10.2020 18:01

Social Studies, 14.10.2020 18:01

Chemistry, 14.10.2020 18:01

Mathematics, 14.10.2020 18:01

Computers and Technology, 14.10.2020 18:01

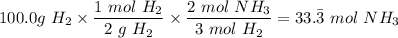
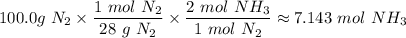
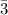 moles of NH₃)
moles of NH₃)

