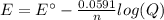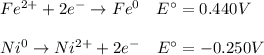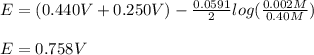Chemistry, 25.11.2020 03:10 zitterkoph
An electrochemical cell is composed of pure nickel and pure iron electrodes immersed in solutions of their divalent ions at room temperature (25°C). If the concentrations of Ni2+ and Fe2+ ions are 0.002 M and 0.40 M, respectively, what voltage is generated at 25°C? (The respective standard reduction potentials for Ni and Fe are −0.250 V and −0.440 V.)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, jmanrules200
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1


Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 12:00, BakerElsie02
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1
You know the right answer?
An electrochemical cell is composed of pure nickel and pure iron electrodes immersed in solutions of...
Questions in other subjects: