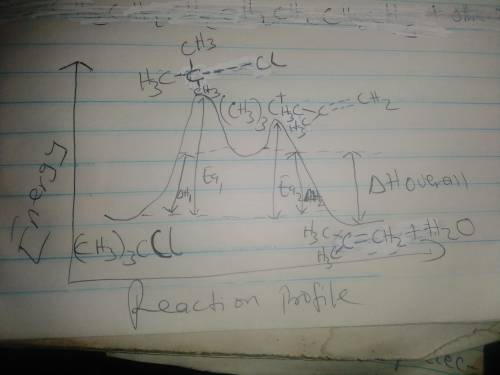
The conversion of (CH3)3CI to (CH3)2C=CH2 can occur
by either one-step or two-step mechanism, as shown in
Equations (1) and (2]
[1]
있
+ I + H₂O
HK
OH
[2]
07
Slow
+ H₂O
+ I
f) Assume Equation [a] represents an endothermic reaction
and that the product of the rate determining step is
higher in energy than the reactants or products.
Draw an energy dagram for this two-step reaction.
Label the axes, reactants and products for each step,
and the Ea and 4to for each step. Label 44° overall.
Draw the structure for both transition states.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, Brookwiggington8814
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 13:30, richardwalker8ourhg2
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a. the mitochondria b. the nucleus c. the vacuoles d. the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 14:40, elawnnalewis4855
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2
You know the right answer?
The conversion of (CH3)3CI to (CH3)2C=CH2 can occur
by either one-step or two-step mechanism, as sh...
Questions in other subjects:


English, 09.12.2019 22:31

Biology, 09.12.2019 22:31

Biology, 09.12.2019 22:31

Mathematics, 09.12.2019 22:31

Biology, 09.12.2019 22:31

Geography, 09.12.2019 22:31



English, 09.12.2019 22:31