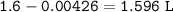Chemistry, 23.11.2020 09:00 Savadt2810
There is a concentrated (12.0 M) solution of HCl in the lab storage area. You need to prepare 1600. mL of HCl solution that has a pH equal to 1.50.
a) What volume of the 12.0 M HCl solution do you need?
b) What volume of water do you need?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, tntaylor862
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1

Chemistry, 22.06.2019 12:00, carvajalj2520
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 22:30, safiyabrowne7594
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
There is a concentrated (12.0 M) solution of HCl in the lab storage area. You need to prepare 1600....
Questions in other subjects:

Computers and Technology, 17.04.2020 05:54



Mathematics, 17.04.2020 06:03

History, 17.04.2020 06:03

Biology, 17.04.2020 06:04

Mathematics, 17.04.2020 06:04


History, 17.04.2020 06:04

Mathematics, 17.04.2020 06:04

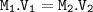
![\tt 10^{-1.5}(pH=-log[H^+])](/tpl/images/0923/0841/24ceb.png) =0.032
=0.032