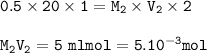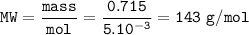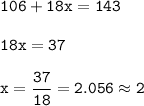Chemistry, 21.11.2020 15:10 esmeraldavelez63
0.715 g of Na2CO3 xH2O is required 20mL of semi-normal hydrochloric acid solution for complete reaction. Find the value of x

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, dgadam7495
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 22:00, choatefarmsus
Does the number of ions in solution increase, decrease, or remain constant? it continuously decreases. it continuously increases. it decreases at first, then increases. it increases at first, then decreases.
Answers: 3

Chemistry, 23.06.2019 03:30, antoinetteee03
Name atleast 3 type of energy associated with the microwave
Answers: 1

Chemistry, 23.06.2019 06:40, hackman1216
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants. thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction. ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch, are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
0.715 g of Na2CO3 xH2O is required 20mL of semi-normal hydrochloric acid solution
for complete reac...
Questions in other subjects:

Mathematics, 23.06.2020 21:01


English, 23.06.2020 21:01


History, 23.06.2020 21:01

English, 23.06.2020 21:01