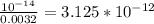Chemistry, 20.11.2020 14:00 sunshine52577oyeor9
Calculate the pH of the resulting solution if 32.0 mL of 0.320 M HCl(aq) is added to (a) 42.0 mL of 0.320 M NaOH(aq). (b) 22.0 mL of 0.420 M NaOH(aq).

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 17:50, mytymikey123
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

You know the right answer?
Calculate the pH of the resulting solution if 32.0 mL of 0.320 M HCl(aq) is added to (a) 42.0 mL of...
Questions in other subjects:


History, 22.08.2019 19:00



Mathematics, 22.08.2019 19:00


History, 22.08.2019 19:00

Mathematics, 22.08.2019 19:00

History, 22.08.2019 19:00

Mathematics, 22.08.2019 19:00