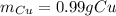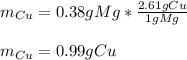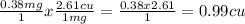Chemistry, 18.11.2020 23:10 queenkimm26
Copper atoms are heavier than magnesium atoms. So, although each atom of magnesium can produce one atom of copper, the masses won’t be the same. The ratio of the atomic weight of copper to the atomic weight of magnesium is about 2.61. Given this ratio and the initial mass of the magnesium strip (0.38 g) measured in task 1, part A, calculate the mass of copper that can be produced. Recall: Mg + Cu(NO3)2 → Cu + Mg(NO3)2

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:40, babygirlqueen5588
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration?
Answers: 3



Chemistry, 22.06.2019 11:50, hamidaakter936848
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2
You know the right answer?
Copper atoms are heavier than magnesium atoms. So, although each atom of magnesium can produce one a...
Questions in other subjects:

History, 07.10.2020 17:01


Mathematics, 07.10.2020 17:01



Biology, 07.10.2020 17:01

History, 07.10.2020 17:01


Mathematics, 07.10.2020 17:01

Mathematics, 07.10.2020 17:01