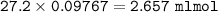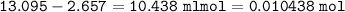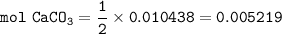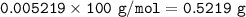Chemistry, 18.11.2020 21:30 lilpeepxliltracy
1.3188g of antacid is weighed and mixed with 75.00 mL of excess 0.1746 M HCl. The excess wcid required 27.20 mL of 0.09767 M NaOH for back titration. Calculate the amount of CaCO3 in the tablet.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, momof7hardings
When would a bouncy ball have the most potential energy
Answers: 2

Chemistry, 22.06.2019 06:30, luhmimi17
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
You know the right answer?
1.3188g of antacid is weighed and mixed with 75.00 mL of excess 0.1746 M HCl. The excess wcid requir...
Questions in other subjects:

Mathematics, 05.10.2019 18:30


History, 05.10.2019 18:30

French, 05.10.2019 18:30


Social Studies, 05.10.2019 18:30

Mathematics, 05.10.2019 18:30

English, 05.10.2019 18:30

Mathematics, 05.10.2019 18:30