
Chemistry, 18.11.2020 21:10 juanaaraujo1104
Dry ice is carbon dioxide in the solid state. 1.28 grams of dry ice is placed in a 5.00 L chamber that is maintained at 35.1°C. What is the pressure in the chamber after all of the dry ice has sublimed? Assume ideal gas behavior.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 07:00, uniqueray33
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

You know the right answer?
Dry ice is carbon dioxide in the solid state. 1.28 grams of dry ice is placed in a 5.00 L chamber th...
Questions in other subjects:

Mathematics, 20.11.2020 05:40

Mathematics, 20.11.2020 05:40



Mathematics, 20.11.2020 05:40


Medicine, 20.11.2020 05:40


Mathematics, 20.11.2020 05:40

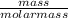
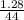 = 0.03moles
= 0.03moles 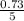 = 0.15atm
= 0.15atm 


