
Chemistry, 18.11.2020 16:50 Fireburntbudder
PLS HELPPP How many moles of oxygen atoms (not molecules) are present in 6.41×1025 molecules of dinitrogen pentoxide (N2O5)? 1. 532 mol 2. 106 mol 3. 32 × 1025 mol 4. 266 mol

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 21:40, fatherbamboo
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 13:30, jcastronakaya
The zinc within a copper-plated penny dissolves in hydrochloric acid if the copper coating is filed down in several spots (so that the hydrochloric acid can reach the zinc). the reaction between the acid and the zinc 2h+(aq)+zn(s)→h2(g)+zn2+(aq) . when the zinc in a certain penny dissolves, the total volume of gas collected over water at 25 °c is 0.947 l at a total pressure of 743 mmhg . (vapor pressure of water is 23.78 mmhg at 25 °c .) what mass of hydrogen gas is collected? answer in appropriate significant figures
Answers: 3

Chemistry, 23.06.2019 16:00, devenybates
Me with this homework my gradebook are closing today so
Answers: 1
You know the right answer?
PLS HELPPP How many moles of oxygen atoms (not molecules) are present in 6.41×1025 molecules of dini...
Questions in other subjects:



Mathematics, 10.03.2021 01:00


Mathematics, 10.03.2021 01:00




Biology, 10.03.2021 01:00

Mathematics, 10.03.2021 01:00

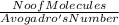
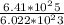
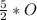
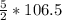 = 266.25 mole
= 266.25 mole

