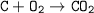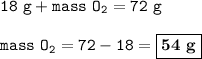Chemistry, 16.11.2020 19:20 Neishab932
In the reaction C + 02 - CO2, 18 g of carbon react with oxygen to produce 72 g of carbon dioxide. What mass of oxygen would be needed in the reaction? O 18 g O 54 g O 72 g 0 90 g

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, PSBSolarYT
For which one of the following reactions is the value of δh° rxn equal to δh° f for the product? a. 2 h2 (g) + o2 (g) → 2 h2o (l) b. n2 (g) + o2 (g) → 2 no (g) c. 2 h2 (g) + o2 (g) → 2 h2o (g) d. h2o (l) + 1/2 o2 (g) → h2o2 (l) e. none of the above
Answers: 1


Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 23.06.2019 05:00, jayden6467
How many moles are in 7.2 x 10^23 carbon molecules?
Answers: 1
You know the right answer?
In the reaction C + 02 - CO2, 18 g of carbon react with oxygen to produce 72 g of carbon dioxide. Wh...
Questions in other subjects:


Social Studies, 23.07.2019 11:30

Physics, 23.07.2019 11:30




Health, 23.07.2019 11:30

Chemistry, 23.07.2019 11:30

Social Studies, 23.07.2019 11:30

Mathematics, 23.07.2019 11:30