
Chemistry, 14.11.2020 01:00 angelasnipes51orfryq
Which is produced in a synthesis reaction?
A) a single element
B) single compound
C) single ion
D) single atom

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, bigwaYne
Imagine that twenty i. u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 11:30, charles8527
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2
You know the right answer?
Which is produced in a synthesis reaction?
A) a single element
B) single compound
C) si...
B) single compound
C) si...
Questions in other subjects:






English, 11.03.2022 01:00




Biology, 11.03.2022 01:00

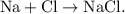 The parent reaction for a synthesis reaction is
The parent reaction for a synthesis reaction is 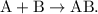
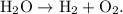 The parent reaction for a decomposition reaction is
The parent reaction for a decomposition reaction is 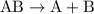 .
.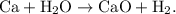 The parent reaction for a single-replacement reaction is
The parent reaction for a single-replacement reaction is 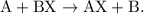
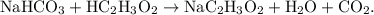 The parent reaction for a double-replacement reaction is
The parent reaction for a double-replacement reaction is 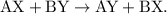
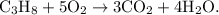 The parent reaction for a combustion reaction is
The parent reaction for a combustion reaction is 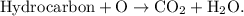 A hydrocarbon is a substance that is only hydrogen and carbon.
A hydrocarbon is a substance that is only hydrogen and carbon.

