
Chemistry, 13.11.2020 06:20 msmojangles
Assume a density of water of 1.00 g/mL, and calculate the mass of water in the solution

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, monithebtslover01
Find the empirical formula of each of the following compounds. given mass or for each element in a sample of the compound 3,611 g ca; 6.389 g c1
Answers: 1

Chemistry, 21.06.2019 18:00, bbombard21
Select the atomic models that belong to the same element
Answers: 2

Chemistry, 22.06.2019 01:00, bettybales1986
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 02:40, alexandraparava
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2
You know the right answer?
Assume a density of water of 1.00 g/mL, and calculate the mass of water in the solution...
Questions in other subjects:

English, 03.06.2021 21:00


Chemistry, 03.06.2021 21:00

Chemistry, 03.06.2021 21:00


History, 03.06.2021 21:00

Mathematics, 03.06.2021 21:00



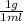 so from this equation, you will get 1 g and you can to SI to be
so from this equation, you will get 1 g and you can to SI to be 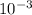 kg
kg

