
Chemistry, 12.11.2020 23:00 leomessifanboy678
Three friends were kicking soccer balls at a neighborhood field. Three soccer balls were rolling across the field. The black soccer ball has less mass than the blue and orange balls, which have equal mass. All three soccer balls were kicked by one of the friends, but not all from the same direction. Use the information in the diagram to answer.
Which soccer ball(s) experienced the strongest force? How do you know?
A. All three soccer balls experienced the same force because they changed speed by the same amount.
B the orange soccer ball it has more mass and the fastest ending speed
c. the blue soccer ball it takes a stronger force to slow down a more massive object than to speed it up.
D the blue and orange soccer balls they have more mass than the black soccer ball changed speed by the same amount
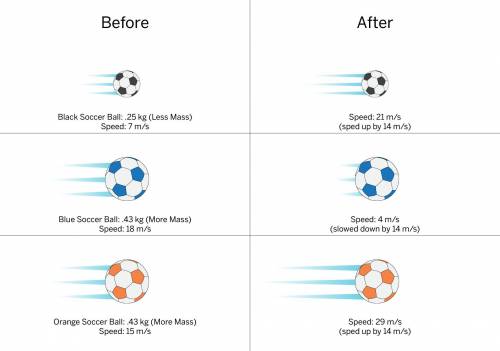

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:50, revlonknox6
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2


Chemistry, 22.06.2019 21:40, k3rbycalilung
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Three friends were kicking soccer balls at a neighborhood field. Three soccer balls were rolling acr...
Questions in other subjects:



Mathematics, 03.12.2020 01:00





English, 03.12.2020 01:00

Mathematics, 03.12.2020 01:00



