
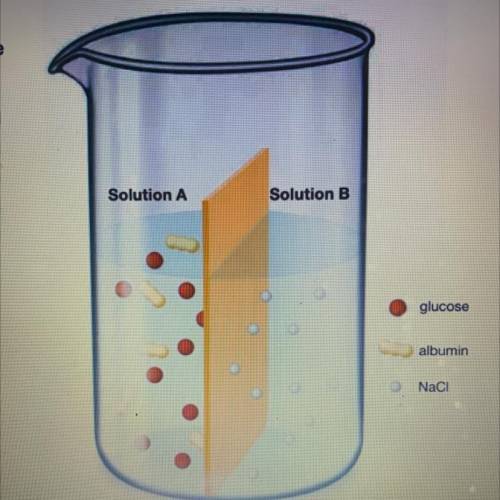
Consider two solutions separated by a semipermeable membrane, as shown in the illustration to the right. The membrane allows the passage of small molecules and ions, but not large molecules like polysaccharides or proteins. Solution A contains a 10% solution composed
of glucose and the protein albumin dissolved in water.
Solution B contains a 5% solution of NaCl in water. Indicate whether each substance in the system would flow into Solution A, Solution B, or neither.
1. Water
2. NaCl
3. glucose
4. Albumin
5. Glucose

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, kichensides
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 10:30, esnyderquintero
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 13:00, nadiarose6345
In a copper wire, a temperature increase is the result of which of the following
Answers: 1

Chemistry, 22.06.2019 22:10, steven0448
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
You know the right answer?
Consider two solutions separated by a semipermeable membrane, as shown in the illustration to the ri...
Questions in other subjects:









Mathematics, 29.08.2019 20:30



