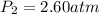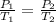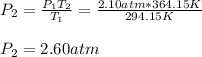Chemistry, 12.11.2020 18:30 elawnnalewis7486
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ∘C. What would the pressure be if the container was heated to 91 ∘C?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, cutebab4786
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3


Chemistry, 22.06.2019 09:30, jewelz5887
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1
You know the right answer?
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ∘C. What would the pressure be if...
Questions in other subjects:


Biology, 01.08.2019 09:30


World Languages, 01.08.2019 09:30


Chemistry, 01.08.2019 09:30

History, 01.08.2019 09:30

Mathematics, 01.08.2019 09:30

History, 01.08.2019 09:30