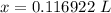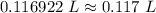Chemistry, 12.11.2020 05:30 PONBallfordM89
Calculate the volume in milliliters of 1.57 M potassium hydroxide that contains 10.3 g of solute.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 05:10, hadellolo8839
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1


Chemistry, 22.06.2019 19:00, HaydenSturgis1
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3
You know the right answer?
Calculate the volume in milliliters of 1.57 M potassium hydroxide that contains 10.3 g of solute....
Questions in other subjects:

Chemistry, 26.09.2019 13:20






Mathematics, 26.09.2019 13:20

History, 26.09.2019 13:20


Social Studies, 26.09.2019 13:20

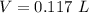
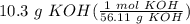 = 0.183568 mol KOH
= 0.183568 mol KOH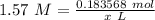 Move x:
Move x: 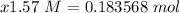 Isolate x:
Isolate x: 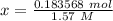 Evaluate:
Evaluate: