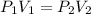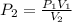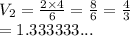Chemistry, 11.11.2020 23:20 flores1717
At a constant temperature, 2L of a gas at 4atm of pressure is expanded to 6L. What is the new pressure? Please show steps!!

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, 10040813
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 14:40, sugardime
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2


Chemistry, 23.06.2019 00:30, runninglovexoxo
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
At a constant temperature, 2L of a gas at 4atm of pressure is expanded to 6L. What is the new pressu...
Questions in other subjects:




English, 10.07.2019 23:30


Mathematics, 10.07.2019 23:30



Chemistry, 10.07.2019 23:30

Biology, 10.07.2019 23:30