

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 14:30, clemsongirl5392
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
What is the frequency of a wave of intrared light which has a wavelength of 4.35 * 10 ^ - 5 meters?...
Questions in other subjects:

History, 15.06.2021 23:50



Chemistry, 15.06.2021 23:50

Mathematics, 15.06.2021 23:50

Mathematics, 15.06.2021 23:50

Biology, 15.06.2021 23:50


Mathematics, 15.06.2021 23:50

Mathematics, 15.06.2021 23:50

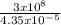 = 6.9 x 10¹²Hz
= 6.9 x 10¹²Hz

