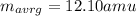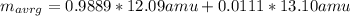Chemistry, 03.11.2020 17:00 mooredollie
An element has two common isotopes. 98.89% of its atoms have an atomic mass of 12.09 amu, whereas the other 1.11% have an atomic mass of 13.10 amu. Using the isotopic composition provided, calculate the average atomic mass of the element.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 18:50, emily9656
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3
You know the right answer?
An element has two common isotopes. 98.89% of its atoms have an atomic mass of 12.09 amu, whereas th...
Questions in other subjects:


Mathematics, 05.02.2020 10:48



Mathematics, 05.02.2020 10:48


History, 05.02.2020 10:48



English, 05.02.2020 10:48