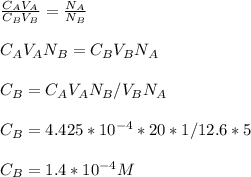Chemistry, 03.11.2020 03:20 sihamabdalla591
3.47 g of the hydrated "double salt", ammonium iron (II) sulfate hexahydrate, FeSO4(NH4)2SO4*6H2O was dissolved in 200. mL of water. 20.0 mL of the solution had some acid added to it and then it reacted completely with 12.6 mL of KMnO4 solution. Calculate the concentration of the KMnO4 solution given the full REDOX equation below. 5Fe2+ + MnO4- + 8H+ --> 5Fe3+ +Mn2+ + 4H2O

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 01:30, arodavoarodavo
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3


You know the right answer?
3.47 g of the hydrated "double salt", ammonium iron (II) sulfate hexahydrate, FeSO4(NH4)2SO4*6H2O wa...
Questions in other subjects:



Mathematics, 26.04.2021 02:50

Mathematics, 26.04.2021 02:50

History, 26.04.2021 02:50

Biology, 26.04.2021 02:50

Mathematics, 26.04.2021 02:50


Social Studies, 26.04.2021 02:50

Mathematics, 26.04.2021 02:50

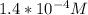
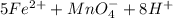 →
→ 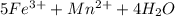
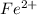 in FeSO₄(NH₄)₂SO₄*6H₂O
in FeSO₄(NH₄)₂SO₄*6H₂O 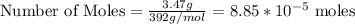
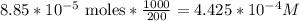
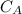 be concentration of
be concentration of 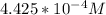
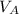 )= 20.0 ml
)= 20.0 ml
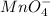 be
be 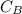 (the unknown)
(the unknown)
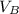 ) = 12.6 ml
) = 12.6 ml
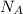 = 5 moles
= 5 moles
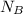 = 1 mole
= 1 mole