
Chemistry, 02.11.2020 21:00 SoccerdudeDylan
An aqueous magnesium chloride solution is made by dissolving 7.15 moles of MgCl2 in sufficient water so that the final volume of the solution is 2.50 L . Calculate the molarity of the MgCl2 solution.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

You know the right answer?
An aqueous magnesium chloride solution is made by dissolving 7.15 moles of MgCl2 in sufficient water...
Questions in other subjects:

Biology, 28.03.2020 01:43

Mathematics, 28.03.2020 01:43

Mathematics, 28.03.2020 01:43



Chemistry, 28.03.2020 01:43




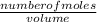
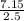 = 2.86mol/L
= 2.86mol/L

