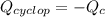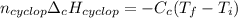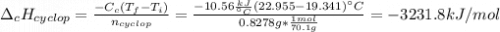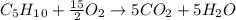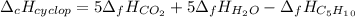Chemistry, 02.11.2020 16:50 uberagentkenny
Cyclopentene is a cyclic hydrocarbon like the ones used in the experiment. In another bomb calorimetry experiment 0.8278 g of cyclopentene is burned and the temperature of the calorimeter increased from 19.341C to 22.955C. The heat capacity of the calorimeter is 10.56 kJ C−1. Calculate the enthalpy of formation of cyclopentene in kJ/mol cyclopentene. Compare this to the accepted value of +32.6 kJ mol−1.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:40, wanderer3653
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 20:30, kittybatch345
Is a chemical message sent by another individual.
Answers: 1
You know the right answer?
Cyclopentene is a cyclic hydrocarbon like the ones used in the experiment. In another bomb calorimet...
Questions in other subjects:


History, 24.08.2019 14:10

Spanish, 24.08.2019 14:10




Physics, 24.08.2019 14:10

Mathematics, 24.08.2019 14:10

History, 24.08.2019 14:10

History, 24.08.2019 14:10