
Based on these descriptions, write a balanced equation and the corresponding Kc expression for each reversible reaction.
a. Carbonyl fluoride, COF2(g), decomposes into gaseous carbon dioxide and gaseous carbon tetrafluoride.
b. Copper metal displaces Silver(I) Ion from aqueous solution , producing silver metal and an aqueous solution of copper(II) ion.
c. Peroxodisulfate ion , S2O82- , oxides iron (II) ion to iron (III) ion in aqueous solution and it is itself reduced to sulfate ion.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, mimithurmond03
The speed of light is around 6.706×10^8 miles per hour. what is the speed of light in units of miles per minute?
Answers: 2

Chemistry, 22.06.2019 03:00, annafellows
Match term definition ellipse a) diagonal cross section of a cylinder circle b) diagonal cross section through the widest part of a sphere sphere c) cross section parallel to the base of a cone great circle d) shape created when a semi-circle is rotated around the y-axis triangle e) perpendicular cross section of a cone
Answers: 1

Chemistry, 22.06.2019 04:30, ajsoccer1705
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3

Chemistry, 22.06.2019 11:50, tajanaewilliams77
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3
You know the right answer?
Based on these descriptions, write a balanced equation and the corresponding Kc expression for each...
Questions in other subjects:


Mathematics, 05.05.2020 16:31




Mathematics, 05.05.2020 16:31


Mathematics, 05.05.2020 16:31


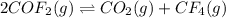
![Kc=\frac{[CO_2][CF_4]}{[COF_2]^2}](/tpl/images/0851/4259/5ff05.png)
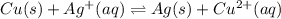


![Kc=\frac{[Fe^{3+}]^2[(SO_4)^{2-}]^2}{[(S_2O_8)^{2-}][Fe^{2+}]^2}](/tpl/images/0851/4259/f3469.png)


