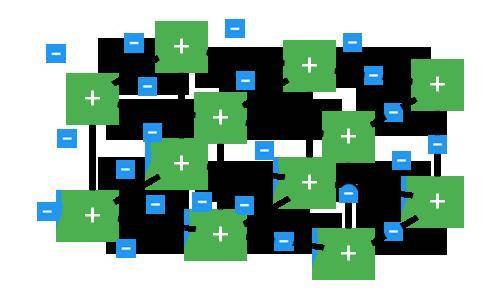
Which type of compound is represented in the model and why?
Select all that apply.
A. T...

Which type of compound is represented in the model and why?
Select all that apply.
A. This model is not a metallic compound because the electrons are being transferred between different ions in the crystal structure.
B. This model is an ionic compound because the rigid formation is an example of a crystal lattice.
C. This model is a metallic compound because it consists of free-moving electrons in a crystal formation.
D. This model is not an ionic compound because the electrons are not being transferred between positive and negative ions; instead, only positive ions are present.

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:50, aesthetickait
How does the motion of particles in a gas change as the gas cools
Answers: 2

You know the right answer?
Questions in other subjects:


Mathematics, 12.11.2020 06:50

Mathematics, 12.11.2020 06:50

Social Studies, 12.11.2020 06:50

History, 12.11.2020 06:50

Mathematics, 12.11.2020 06:50


Biology, 12.11.2020 06:50

Mathematics, 12.11.2020 06:50

English, 12.11.2020 06:50



