
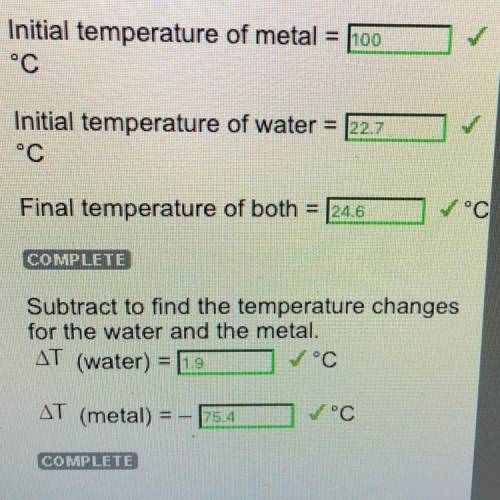
Step 7: Put the Metal in the water and Measure Temperature Changes (Copper) Initial temperature of metal
= 100°c
Initial temperature of water = 22.7°C
Final temperature of both
24.6°C
Subtract to find the temperature changes
for the water and the metal.
AT (water) = 1.9
AT (metal) = -75.4

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, david838843
Iwll give extra points to who gets this for ! what type of reaction is this? ?
Answers: 2

Chemistry, 21.06.2019 21:30, cicimarie2018
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 23.06.2019 04:31, laurenbreellamerritt
How big are the bighest ocean waves at mavericks
Answers: 1

Chemistry, 23.06.2019 15:30, georgesarkes12
Floor 19 is an isotope of fluorine it has different number a. electrons b. protons c. neutrons d. electron shells
Answers: 1
You know the right answer?
Step 7: Put the Metal in the water and Measure Temperature Changes (Copper) Initial temperature of m...
Questions in other subjects:

Mathematics, 10.11.2020 08:00


Chemistry, 10.11.2020 08:00

Spanish, 10.11.2020 08:00

Mathematics, 10.11.2020 08:00

English, 10.11.2020 08:00



History, 10.11.2020 08:00

English, 10.11.2020 08:00



