
Chemistry, 23.10.2020 22:20 yoangel9183
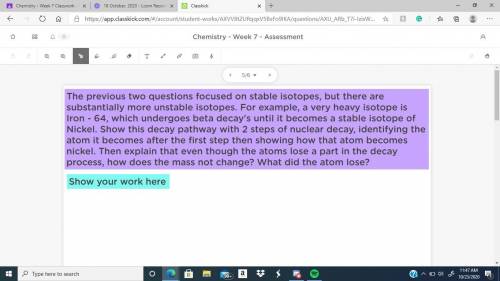
the previous 2 questions focused on stable isotopes, but there are substantially more unstable isotopes. For example, a very heavy isotope is iron-64, which undergoes beta decay until it becomes a stable isotope of nickel. Show this decay pathway with 2 steps of nuclear decay, identifying the nickel. Then explain that even though the atoms lose a part in the decay process, how does the mass not change? what did the atom lose?

Answers: 1


Other questions on the subject: Chemistry



You know the right answer?
the previous 2 questions focused on stable isotopes, but there are substantially more unstable isoto...
Questions in other subjects:






Chemistry, 22.06.2019 07:30

History, 22.06.2019 07:30

Chemistry, 22.06.2019 07:30




