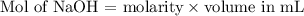You will perform several calculations with the data you obtain in the titrations lab. Below is an exercise to help you prepare. (a) If your titration solution is 0.450 M in NaOH, and the endpoint occurs at 13.70 mL of titrant, how many mmol of NaOH are required to reach the endpoint

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1


Chemistry, 22.06.2019 13:30, nasibamurodova
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1
You know the right answer?
You will perform several calculations with the data you obtain in the titrations lab. Below is an ex...
Questions in other subjects:

Mathematics, 30.11.2020 17:40

Business, 30.11.2020 17:40

Mathematics, 30.11.2020 17:40






Physics, 30.11.2020 17:40