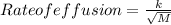Chemistry, 22.10.2020 19:01 juliannabartra
At constant temperature, He effuses 6.04 times faster than a gas having an unknown molar mass. Determine the molar mass of the unknown gas.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 01:50, lildestinyquintana
Ase your answer to this question on the information below. hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant. the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen. chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product. nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
You know the right answer?
At constant temperature, He effuses 6.04 times faster than a gas having an unknown molar mass. Deter...
Questions in other subjects:

Mathematics, 21.10.2019 17:30

English, 21.10.2019 17:30



Spanish, 21.10.2019 17:40

Mathematics, 21.10.2019 17:40

Business, 21.10.2019 17:40


Mathematics, 21.10.2019 17:40

Mathematics, 21.10.2019 17:40