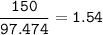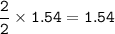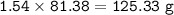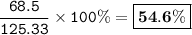Chemistry, 22.10.2020 05:01 passions3534ovf6dt
When 150. g zinc sulfide are burned in excess oxygen, 68.5 g of zinc oxide are actually produced, along with sulfur dioxide. Determine the percent yield of zinc oxide after finding the theoretical production of zinc oxide. **Show the balanced equation before you begin.**

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:10, rightstrong9827
When the volume and number of particles of a gas are constant which of the following is also constant
Answers: 3


Chemistry, 22.06.2019 09:30, lisbet123085
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
When 150. g zinc sulfide are burned in excess oxygen, 68.5 g of zinc oxide are actually produced, al...
Questions in other subjects:


History, 18.12.2019 07:31




English, 18.12.2019 07:31


Physics, 18.12.2019 07:31


Social Studies, 18.12.2019 07:31