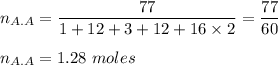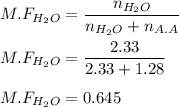Chemistry, 21.10.2020 16:01 babyash12718
A solution is made by mixing of 42.g water and 77.g of acetic acid HCH3CO2. Calculate the mole fraction of water in this solution.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1



Chemistry, 22.06.2019 15:00, kandi2565
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2
You know the right answer?
A solution is made by mixing of 42.g water and 77.g of acetic acid HCH3CO2. Calculate the mole fract...
Questions in other subjects:





English, 11.04.2020 17:55

History, 11.04.2020 17:55

Mathematics, 11.04.2020 17:55



Geography, 11.04.2020 17:55

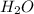 ,
,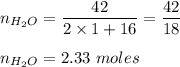
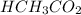 ,
,