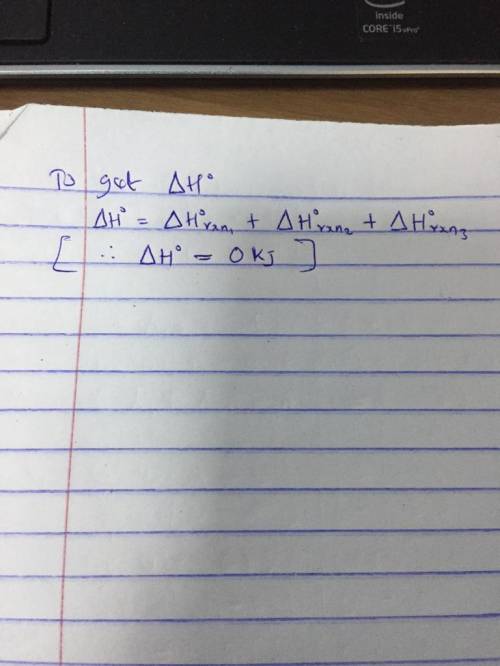6.82
A 2.10-mole sample of crystalline acetic acid, ini-
tially at 17.0°C, is allowed t...

6.82
A 2.10-mole sample of crystalline acetic acid, ini-
tially at 17.0°C, is allowed to melt at 17.0°C and is
then heated to 118.1°C (its normal boiling point) at
1.00 atm. The sample is allowed to vaporize at.
118.1°C and is then rapidly quenched to 17.0°C, so
that it recrystallizes. Calculate AH° for the total pro-
cess as described.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, lorenaandreahjimenez
The answer for #3 is c but i don't know why
Answers: 1

Chemistry, 22.06.2019 04:30, homeschool0123
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 10:00, alexabdercmur
Why is the structure of molecule important to its function?
Answers: 1
You know the right answer?
Questions in other subjects:

History, 21.07.2019 15:00



History, 21.07.2019 15:00


Health, 21.07.2019 15:00

History, 21.07.2019 15:00


Biology, 21.07.2019 15:00

Health, 21.07.2019 15:00